MARINE SCIENCE: VIRTUAL URCHiN LAB ( OCEAN ACIDIFICATION)
Essential Question: The ocean covers 2/3 of the planet- Is it really
possible that humans are changing the chemistry of the ocean?
NO
1: Carbon in the Air: What does the graph of atmospheric Carbon
Dioxide tell us?
This graph shows atmospheric CO2 measurements ( y-axis) over a 50 year time span ( x- axis).
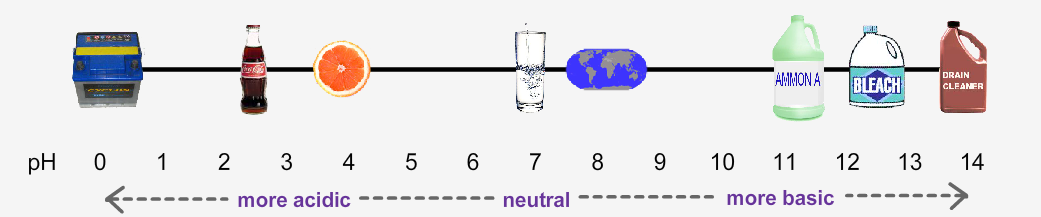
2: pH Scale: Where do some common items fall on the pH scale? Take a screen shot of your
final answers and paste below:
some common items fall on the left on the ph scale (more acidic< neutral)
possible that humans are changing the chemistry of the ocean?
NO
1: Carbon in the Air: What does the graph of atmospheric Carbon
Dioxide tell us?
This graph shows atmospheric CO2 measurements ( y-axis) over a 50 year time span ( x- axis).
2: pH Scale: Where do some common items fall on the pH scale? Take a screen shot of your
final answers and paste below:
some common items fall on the left on the ph scale (more acidic< neutral)
3: Ocean pH: How might it change? A change from 8.2 to 8.1 on the pH scale is a 30%
increase in acidity.
4: Carbon in the Water: Explain what happens to Carbon in ocean water. The continuing buildup of carbon dioxide (CO2) in the atmosphere means more CO2 going into
the oceans. Carbon dioxide dissolves in seawater to form carbonic acid (H2CO3).
5: Exploring Carbon Levels and Effects: Look over the interactive and describe what happens
at each of the levels:
Scenario 1: Optimistic
-Every ten years the pH of the ocean decreases .2%
Scenario 2: Middle Ground
-The carbon dioxide in the ocean increases.
Scenario 3: Pessimistic
-Every ten years the pH of the ocean water decreases by .2% and the carbon dioxide increases at least 29 more.
6: Diversity of Life in the Sea: Sort each of the organisms into Calcifiers or Non-Calcifiers.
Take a screenshot of your final answer and paste below:
increase in acidity.
4: Carbon in the Water: Explain what happens to Carbon in ocean water. The continuing buildup of carbon dioxide (CO2) in the atmosphere means more CO2 going into
the oceans. Carbon dioxide dissolves in seawater to form carbonic acid (H2CO3).
5: Exploring Carbon Levels and Effects: Look over the interactive and describe what happens
at each of the levels:
Scenario 1: Optimistic
-Every ten years the pH of the ocean decreases .2%
Scenario 2: Middle Ground
-The carbon dioxide in the ocean increases.
Scenario 3: Pessimistic
-Every ten years the pH of the ocean water decreases by .2% and the carbon dioxide increases at least 29 more.
6: Diversity of Life in the Sea: Sort each of the organisms into Calcifiers or Non-Calcifiers.
Take a screenshot of your final answer and paste below:
7: Echinoderms Life Cycles and Skeletons: Describe the life cycle of Echinoderms and explain why they are so sensitive to changes in pH.
at first they start as larva then they grow becoming of hat its called urchin lava. After this larva starts groing it start developing some juvenile structures, and right after the skeleton developed to its mature style it becomes of what we call methamorphosis.
8: How to Study Ocean Acidification in the Lab- How may ocean acidification impact Sea Urchins in the larval stage? Can you think of any other possible acidification impacts on marine organisms other than calcification?
It make the ocean more acidic , which makes more difficult to these larva to develop their skeleton.
Can you think of any other possible acidification impacts on marine organisms other than calcification?
Another factor that may affect the ocean acidification can be the dirty waters that industries throw into the ocean.
Welcome to the Ocean Acidification Lab!
1: Complete the pre-lab certification.
2: Look at the first slide under the microscope- Describe what you see.
are using these two pH values for this experiment.
at first they start as larva then they grow becoming of hat its called urchin lava. After this larva starts groing it start developing some juvenile structures, and right after the skeleton developed to its mature style it becomes of what we call methamorphosis.
8: How to Study Ocean Acidification in the Lab- How may ocean acidification impact Sea Urchins in the larval stage? Can you think of any other possible acidification impacts on marine organisms other than calcification?
It make the ocean more acidic , which makes more difficult to these larva to develop their skeleton.
Can you think of any other possible acidification impacts on marine organisms other than calcification?
Another factor that may affect the ocean acidification can be the dirty waters that industries throw into the ocean.
Welcome to the Ocean Acidification Lab!
1: Complete the pre-lab certification.
2: Look at the first slide under the microscope- Describe what you see.
- a larve in circle form , it also turns to normal light, ere you can barely see it it.
are using these two pH values for this experiment.
- We are using to different kinds of pH's so then e can see the differences of these two different factors. Also because the pH of ocean water is 8.1 while the 7.7 ph can be the the average ph for an end of a century.
- the ph increases